Many scientists working tirelessly around the world have concluded that the world’s most popular herbicide is actually a serious dangerous to health. This painstaking study from Europe should cast no doubt on the seriousness of being exposed.
Glyphosate: Too Much of a Good Thing?
- 1Faculty of Health Sciences, Institute of Pharmacology, University of Tromsø, Tromsø, Norway
- 2GenØk – Centre for Biosafety, Tromsø, Norway
- 3Czech Agriculture and Food Inspection Authority, Praha, Czech Republic
Although previously accepted as the less toxic alternative, with low impact on animals, farmers as well as consumers who are exposed to residues in food, glyphosate chemicals are now increasingly controversial as new evidence from research is emerging. We argue that specific aspects of the history, chemistry and safety of glyphosate and glyphosate-based herbicides should be thoroughly considered in present and future re-evaluations of these dominant agrochemicals:
• Glyphosate is not a single chemical, it is a family of compounds with different chemical, physical, and toxicological properties.
• Glyphosate is increasingly recognized as having more profound toxicological effects than assumed from previous assessments.
• Global use of glyphosate is continuously increasing and residues are detected in food, feed, and drinking water. Thus, consumers are increasingly exposed to higher levels of glyphosate residues, and from an increasing number of sources.
• Glyphosate regulation is predominantly still based on primary safety-assessment testing in various indicator organisms. However, archive studies indicate fraud and misbehavior committed by the commercial laboratories providing such research.
We see emerging evidences from studies in test-animals, ecosystems indicators and studies in human health, which justify stricter regulatory measures. This implies revising glyphosate residue definitions and lowering Maximum Residue Limits (MRLs) permissible in biological material intended for food and feed, as well as strengthening environmental criteria such as accepted residue concentrations in surface waters. It seems that although recent research indicates that glyphosates are less harmless than previously assumed and have complex toxicological potential, still regulatory authorities accept industry demands for approving higher levels of these residues in food and feed.
Introduction
In As You Like It by Shakespeare, Rosalind asks Orlando: “Can one desire too much of a good thing?” …
The phytotoxic properties of glyphosate were recognized around 1970 and the new compound was enthusiastically embraced as a good thing; it was perceived as a practically non-toxic alternative, a safe chemical and a benefit to society. And, best of all, it proved to be an efficient herbicide. After introduction of first commercial formulations around 1975, glyphosate-based herbicides (GBHs) have become globally dominant for eradication of unwanted weed species and lately also have found other use, e.g., as desiccants on agriculture crops. At the moment of writing, glyphosate is the globally dominating herbicide, measured in tonnage, and revenue.
Archive film from a commercial biotechnology laboratory in 1987 shows George H. W. Bush (at that time vice-president of the USA) as he asks the assembled researchers; “this gene of yours, what does it do in the plant?” Before any of the superiors have a chance to answer, a junior scientist excitedly proclaims; “we have this fabulous herbicide…” (Robin, 2008). The fabulous herbicide was glyphosate and the gene in question was the commercially promising EPSPS gene isolated from Agrobacterium, which made it possible to modify agriculture crops into glyphosate-tolerant varieties. Leading agronomists later described the fabulous herbicide in a widely acknowledged publication bearing the title; “Glyphosate: a once-in-a century herbicide” (Duke and Powles, 2008). The headline for this present writing also refers to the 2012–2015 detailed evaluation of glyphosate recently completed by the European Food Safety Authority: “Glyphosate: EFSA updates toxicological profile” (EFSA, 2015c), in which EFSA concludes that glyphosate is probably not a human carcinogen, but on the other hand also acknowledges the need for tighter regulation, specifically by adjusting consumer exposure.
GBHs were primarily intended for pre-emergence application in conventional industrial agriculture. However, genetically modified cultivars (GM) allow post-emergence application in herbicide-tolerant genetically modified soybean, corn, cotton, and canola. These crops are engineered to withstand the effects of glyphosate and seen as a main incitement for increasing production of and application of these chemicals (Charles, 2001; Benbrook, 2012, 2016; Bonny, 2014; Cuhra, 2015a). Annual global production figures for glyphosate have recently been estimated at 825,800,000 kg (Benbrook, 2016), while less investigative sources estimate even higher production volumes, surpassing 1 million ton annually (Székács and Darvas, 2012; Bøhn et al., 2016) and there are few indications of reduced use, other than challenges from resistant weed and emerging evidence.
A string of previous studies investigated aspects such as toxicity of glyphosate and Roundup toward aquatic invertebrates (Cuhra et al., 2013), accumulation of glyphosate in glyphosate-tolerant soybean (Bøhn et al., 2014) and potential effects of such residues in test animal feed (Cuhra et al., 2014, 2015). Furthermore, we have reviewed reports from industry studies investigating these issues. No studies other than our own were found to specifically assess effects of glyphosate residues (Cuhra, 2015b). We have also published preliminary results from studies of documentation in archives from US FDA and US EPA, obtained via freedom of information act requests (Cuhra, 2015c).
Importantly, glyphosate is not one single clearly defined compound, but rather a family of chemicals that can be synthesized through different chemical processes, which in turn will cause various qualitative differences e.g., impurities and byproducts. Glyphosates exist in several chemical mixtures and/or forms, primarily as either glyphosate technical acid or as various salts of glyphosate.
Review of Recent Findings
Compositional analysis of soybean samples from major production areas in USA and Argentina determined that such transgenic glyphosate-tolerant crops accumulate glyphosate, causing surprisingly high levels of glyphosate residues (Bøhn et al., 2014), even far above the spacious maximum residue limits which exists for soybean at present (Then, 2013; Cuhra, 2015b). Such high levels of glyphosate residues are endemic for the glyphosate-tolerant GM-varieties. In samples of organic soy and conventionally grown soy from industrial agriculture, no such residues were detected (n = 21, LOQ = 0.1 mg/kg) (Bøhn et al., 2014). Subsequent research indicated that (a) soy from organic agriculture gave better growth, survival and reproduction in the indicator organism Daphnia magna, compared to conventional GM and non-GM varieties of soy (Cuhra et al., 2014). Furthermore, (b) subsequent testing demonstrated that when D. magna was fed diets made with soybean meal from Roundup-ready soybean, the biological parameters growth and reproduction were negatively correlated with the magnitude of glyphosate residues (in a 42-day experiment involving 300 animals allocated to eight separate diets with known glyphosate-residue concentrations, all below existing legal limits) (Cuhra et al., 2015). Furthermore, we found that glyphosate isopropyl amine salt (glyphosate IPA) in water, had 100-300-fold higher acute toxicity toward test-organism D. magna, as compared with industry studies using same species of test-animal and similar methodology of testing. And, in long-term studies we found that low concentrations (0.05–0.45 ppm) of either glyphosate (glyphosate IPA) or GBH Roundup formulation (contains glyphosate IPA as active ingredient) had adverse effects on growth and reproduction (Cuhra et al., 2013). Several problematic issues relating to existing assumptions on ecotoxicity of glyphosate were identified, amongst these the fact that glyphosates are a family of chemicals with distinctly differing physical properties and biological effects, notably levels of contaminants from different manufacturing processes, and basic properties such as solubility in water (Cuhra, 2015c). Also, numerous studies on toxicity and ecotoxicity of glyphosate and glyphosate-based herbicides were performed by commercial laboratories at a time when such research did not adhere to later quality requirements. Thus, it has been concluded that amongst the industry-funded studies providing data for the regulatory basis by documenting glyphosate safety, there are studies which should be reviewed and discarded as evidence of safety (Cuhra, 2015b,c).
The lack of relevant risk-assessment data may come from lack of valid studies, since research commissioned and funded by industry is found to ignore the question of herbicide use as well as residue levels in the plant material, and possible effects from these (Millstone et al., 1999; Viljoen, 2013; Cuhra, 2015b). Millstone et al. documented serious flaws in initial assessments presented by industry as evidence of safety: Most safety assessments had been conducted using herbicide tolerant plant material which was not sprayed with its belonging herbicide, and thus could not have the levels of glyphosate residues which would be expected under normal agriculture practice. Later, Viljoen confirmed this to still be the case in most feeding studies performed to test the quality of herbicide tolerant GM plants. Our recent review of the issue highlights that not only are the test materials for research cultivated in artificial environments, but the question of glyphosate residues continues to be an ignored issue. We argue that such unfortunate gap in knowledge originates in a societal acceptance of industry autonomy and that the responsibility for providing data for safety-assessment studies is delegated to the producer of the product (Cuhra, 2015b). Only one of 30 reviewed studies was found to address the question of glyphosate residues, and that was a compositional study performed by us (Bøhn et al., 2014). Such methodological flaws in industry studies not only discredit and undermine the claimed substantial equivalence of GM cultivars, they also point to an insufficient regulatory oversight over knowledge gaps related to important safety-issues (Cuhra, 2015b).
Glyphosates and formulated GBH products such as Roundup have been subjected to a large number of studies: Researchers have investigated glyphosates and their role in industrialized farming practices, from various scientific disciplines and from a wealth of perspectives; agronomy (Duke and Powles, 2008; Benbrook, 2012, 2016; Bonny, 2011, 2014), socio-economy (Binimelis et al., 2009; Bonny, 2014), Ecology (Giesy et al., 2000; Samsel and Seneff, 2015b) and health (Williams et al., 2000; Samsel and Seneff, 2013, 2015a,b; Mesnage et al., 2015a). Few independent scientists (researchers not employed by industry) have voiced such univocal praise as the agronomists who published the initially mentioned commentary in which glyphosate is stated to be “a once-in-a-century herbicide,” “a precious herbicidal resource” and a “unique ideal herbicide” (Duke and Powles, 2008). Some of these claims seemed justified at the time of writing, especially since these evaluations arose before the more recent; (1) findings of high levels of accumulation in food and feed, (2) findings of destructive outbreaks of glyphosate-resistant weeds, and (3) indications of complex toxic effects.
Hence, although glyphosate was initially found to be environmentally benign, to have low toxicity to farm workers and other non-target organisms, and to be biodegradable, several of these assumptions of the “unique ideal herbicide” have recently been scrutinized and questioned.
In addition, GBHs include a large diversity of herbicidal products, i.e., more than 750 formulated products are found on the market (Guyton et al., 2015), with unknown additive ingredients, making evaluation and testing even more difficult.
At present, the global database at www.weedscience.org has registered 32 different species of weeds tolerant to glyphosate (Heap, 2015). Arguably, the reaction to these recent challenges has partly contributed to increase the ecological challenges: we see that a main strategy applied by agroindustry has been to further develop technical and biological modifications of agriculture crops, in order to facilitate even higher application dosage of glyphosates as active ingredients in products (Cuhra, 2015b). This is increasingly affecting local biota and farming systems as application rates on individual fields increase, in an unsustainable spiraling development which should be evaluated carefully (Binimelis et al., 2009). Another approach is to combine tolerances to several herbicides in the same transgenic plant (Green, 2009).
Chemistry and History of Glyphosates
The common name “glyphosate” is used indiscriminately in published literature, denominating various chemical compounds that differ substantially from the glyphosate-IPA salt (chemical identity CAS# 38641-94-0), e.g., the technical grade glyphosate (CAS# 1071-83-6). Toxicological data for technical grade glyphosate are not relevant when assessing ecological effects of glyphosate herbicides, which contain water-soluble forms of glyphosate, e.g., the IPA-salt, as the active ingredient.
In this context we again find it relevant to highlight the types of glyphosate which are used in agriculture as active chemical ingredient in glyphosate-based herbicides (GBH). These are primarily glyphosate isopropyl amine, glyphosate ammonium, glyphosate sesquisodium, and glyphosate trimesium salts. It is these glyphosate-salts that are the primary glyphosate chemicals released into the environment and which are sources of residues or metabolites subsequently found in various feed- and foodstuff.
Different glyphosate compounds have slightly or profoundly different properties. An overview can be found at the PubChem online database (hosted by the US National Institutes of Health at https://pubchem.ncbi.nlm.nih.gov) presenting a synthesis of information on physical, chemical, and toxicological properties of chemicals. Glyphosates are pooled in Compound identity CID #3496. This entry includes glyphosate technical acid, but also various other glyphosate chemicals such as the isopropylamonium salts (IPA-salts), which are commonly used in commercial herbicides. The PubChem database also provides common synonyms and lists major producers of glyphosate, including a range of different glyphosate chemicals which these producers offer onto the commercial market. Links to hundreds of records on related compounds in the database present confusing information, especially as the commercially and environmentally important glyphosate salts obscurely are also listed in other subdivisions of the database.
Also, we notice that there are several independent systems for nomenclature of chemicals including glyphosates. The PubChem database employs CID-codes for chemical compounds. These are different from the universally recognized CAS-codes. Also, although US EPA documentation on glyphosates refers to CAS-codes, additional codes (e.g., internal codes and “Shaughnessy” codes) are used. Authorities such as the US Department of Labor use an altogether different nomenclature for glyphosate (OSHA–IMIS codes, in which glyphosate-IPA is given the identity “R107”). This diversity of codes results in confusing nomenclature which subsequently complicates scientific assessments and regulatory approvals.
The following examples illustrate the challenges for identifying correct type of glyphosate for testing: For many years (and to some degree still) the US EPA Reregistration Eligibility Decision (RED) on glyphosate (US EPA, 1993) has been the main document on glyphosate in the US administration and an important reference for assessment of potential effects on health and environment. However, the supporting technical dossier (Shaughnessy Case No. 0178) confuses the physical properties of two different main glyphosate chemicals: The IPA-salt specification gives data on melting point, density, and water-solubility. Again, amongst these properties, the water-solubility is most important in a chemical intended to be diluted with water. However, the documentation presents the very low solubility of the glyphosate technical acid (at 10 g/l this is relatively insoluble and not relevant as an active ingredient in commercial formulations, in comparison the IPA-salt has solubility exceeding 1000 g/l). The RED is largely based on data provided by the industry manufacturer of the glyphosate chemicals (partly confidential information protected by national and international patents) and evidently has been compiled without the necessary differentiation between glyphosate forms.
Numerous published experiments on ecotoxicological effects of GBH in various species and environments have tested the glyphosate technical acid (the parent compound). However, studies on effects of glyphosate technical acid are not relevant for assessing the potential effects of the glyphosate active ingredient in herbicides. We argue that this is a possible explanation for the contradictory published results in specific species of test-animals and specific test-systems, presenting EC50 values which span several orders of magnitude (Cuhra et al., 2013).
Furthermore, analysis of glyphosate residues in environmental samples, food and feed, have quantified only “glyphosate” (as N-phosphonomethyl glycine) and the defined main metabolite “AMPA” (aminomethylphosphonic acid). The newest revision of central EFSA documents on glyphosate (EFSA, 2015a) begins to take these questions into account. The document specifies that the IPA-salt of N-phosphonomethyl glycine (glyphosate-IPA) is the relevant compound for assessment and also presents some details on other metabolites (N-acetylglyphosate (NAG), N-acetyl-AMPA), and impurities. As presented in the EFSA document, N-acetylglyphosate, and N-acetyl-AMPA are newly proposed to be part of the residue definition for monitoring and for dietary risk assessment. They occur in certain genetically modified plants such as soybeans or maize following application of glyphosate and were evaluated by EFSA with regard to setting of import tolerances. It was noted that formaldehyde may occur as an impurity and a content of 1 g/kg or higher in the active ingredient would result in a classification as a 1B carcinogen (EFSA, 2015a).
We find that the chemical and biological processes of glyphosate degradation are insufficiently documented and we expect that other potential metabolites and additional residues could also be of importance. Also, the break-down rates of glyphosates are relevant. Glyphosate and AMPA residues in samples of Roundup-ready soybean were analyzed two years after harvest. We found high concentrations of both chemicals (mean 3.3 mg/kg of glyphosate and 5.7 mg/kg of AMPA) (Bøhn et al., 2014), i.e., somewhat more AMPA (63% of the total) than glyphosate. This indicates that in stored seeds, glyphosate degrades slowly.
A classic and somewhat morbid joke states that five out of six scientists conclude, that Russian roulette is safe. The evidence on glyphosate safety is of this nature, as a majority of previous studies (before 2010) find that glyphosate is safe, contrasted by only a minority of studies which find that glyphosate causes harm. Returning to the metaphor of the revolver in the undoubtedly dangerous game of Russian roulette, an inspection would reveal that only one chamber is loaded with a functional cartridge, the others are blanks. Based on our review of published glyphosate safety assessments we conclude that the mentioned metaphor is highly relevant. We see that an important cluster of publications, which can be said to be at the core of evidence demonstrating safety of glyphosate herbicides, was not performed using the relevant type of glyphosate chemical. Thus, those safety assessments investigated “blanks,” whereas a few supplementary studies have tested the actual glyphosate herbicide or the active ingredients correctly representative of the actual chemicals dispersed onto farmlands and into the environment.
We recommend focusing further on the studies which investigate representative glyphosate, instead of concluding from studies that have investigated the parental compounds of glyphosate. Regulatory authorities must be capable to separate real bullets from blanks when assessing evidence for risk-assessment. Only the effects of real bullets are relevant.
Toxicity and Ecotoxicity of Glyphosates and GBHs
Roundup and similar formulated glyphosate herbicides contain various adjuvants and inert ingredients. We have described some of the confusion that enshrouds ecotoxicological and toxicological assessments of these compounds, which are seen as significantly contributing to toxicological properties of formulated herbicides (Cuhra et al., 2013; Cuhra, 2015c). Recognizing the inherent complexity of assessing compounds which are protected commercial products and which have properties known to producers, but partly unavailable to scientists and regulators, we suggest that all ingredients in herbicide formulations should be regulated and subject to mandatory declaration. Present regulation allows producers of formulations to simply declare various additives and adjuvants as “inert ingredients,” although such GBH-compounds were initially recognized to have biological and toxicological effects in non-target organisms (Folmar et al., 1979).
The best-known GBH products are Roundup formulations that contain additional surfactants, chemical adjuvants. Recent papers have reviewed published literature on GBH-formulation toxicity (Mesnage et al., 2014, 2015a). Typically, Roundup contains glyphosate as IPA-salt, polyethoxylated tallow amine (POEA) and additional substances. These adjuvants may in some cases be more toxic than the glyphosate active ingredient itself (Howe et al., 2004; Peixoto, 2005). The phenomenon of potentially higher toxicity in formulated herbicides, as compared to the active ingredient only, is documented for glyphosate-based herbicides as well as for a number of other herbicide active ingredients (Mesnage et al., 2014). Recent evidence indicates that glyphosate has complex toxic effects (Samsel and Seneff, 2015b) and supports the hypothesis that co-formulants to glyphosate in Roundup are endocrine disruptors in human cells (Defarge et al., 2016). Relative to this, our ecotoxicological comparative testing of glyphosate (IPA-salt) and Roundup “Weed & Grass Killer Concentrate Plus” in D. magna, has shown that the active ingredient and the formulated product have approximately the same acute toxicity (short-term), although the formulated product did produce more severe effects in long-term exposure (life-long) (Cuhra et al., 2013).
GBH (Roundup) has been shown to disturb male reproductive systems through Ca2+-mediated toxicity, oxidative stress and disruption of signaling mechanisms in rats (Cavalli et al., 2013). This also happened at concentrations below what farm workers typically are exposed to Cavalli et al. (2013). Further, both acute and chronic exposure to Roundup may cause oxidative stress and neurotoxicity in rats (Cavalli et al., 2013; Cattani et al., 2014), justifying claims of being a neurotoxic hazard also for humans (Malhotra et al., 2010; Grandjean and Landrigan, 2014). Some evidence of arrhythmic and cardiac electrophysiological changes mediated by GBH also indicate cardiovascular risk to animals and humans (Gress et al., 2015).
A recent study investigated gene expression changes in rats after long-term exposure to Roundup at very low concentrations (0.1 μg/kg) in the drinking water. The results showed that 263 genes from kidney and liver had a fold-change >2, indicating liver and kidney damage and potential health implications also in other animals including humans (Mesnage et al., 2015b). Roundup, but not “pure glyphosate” (not clarified what type), was shown to cause endocrine disruption in Leydig cells (Walsh et al., 2000), indicating significant activity in other components of formulations. An additional recent review by Mesnage et al. summarizes further evidence that Roundup at or below regulatory limits may be toxic or cause teratogenic, tumorigenic, and hepatorenal effects (Mesnage et al., 2015a). Such effects can be linked to endocrine disruption and oxidative stress (Gasnier et al., 2009).
Glyphosate Mode-of-Action
The herbicidal properties of glyphosate (N-phosphonomethyl-glycine) inhibit biosynthesis of chorismate from shikimate (Amrhein et al., 1980), thereby lethally disrupting photosynthesis and plant cell metabolism. It has been claimed that since only plants (and some lichens and microorganisms) have the 5-enolpyruvylshikimic acid-3-phosphate synthase metabolic pathway (EPSPS pathway) defined as glyphosate target-site, only such organisms can be expected to be targeted by toxic effects of this chemical (Duke et al., 2012). Arguably, such general deduction of safety toward non-target organisms is scientifically unfounded. It is not justified to assume that specific chemicals have only one mechanism or mode-of-action in ecosystems, biota and species. Toxins can interact with numerous biochemical processes in cells, tissues, and organs of various organisms.
Published Evidence on Glyphosate and Safety
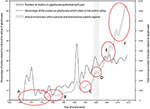
A brief database search on term “glyphosate,” alternatively the term “glyphosate” combined with term “safety” or term “risk” determined by Boolean operator “AND” and “OR” via the Google Scholar search engine yields data presented in Figure 1.
Figure 1. Glyphosate research and development. A: Period of development, approval and patenting. B: Initial safety assessments. C: Advent of glyphosate-tolerant-crops. D: Several important reviews on health and ecotoxicology documenting low toxicity. E: Emerging evidence on crop damage, residue accumulation and impact on non-target organisms. F: Evidence on complex effect.
The total number of peer reviewed scientific articles and related posts such as technical reports and patent documents on “glyphosate” published 1965–2014 (search date 24/09/2015) is found to be 62.200. Using at least one of the terms “safety” or “risk” in addition to “glyphosate” returns 20.900 scientific articles and related posts. These total figures on glyphosate are found to be comparable to the available evidence on herbicide atrazine and insecticides malathion and dieldrin determined by similar searches using same search-terms and conducted in the same period (Table 1).
Table 1. Search results as number of publications 1965–2015 on four pesticide active ingredients (a.i): herbicides glyphosate and atrazine, insecticides dieldrin, and malathion.
The annual total publications on “glyphosate” are visualized in Figure 1. We extracted data for each year from 1970 to 2014, thus covering glyphosate research over 45 years. The quantity of publications on glyphosate rise exponentially (gray line) to the present level of 9.435 registrations in 2014. Although there are some fluctuations in the rates (percentages) of safety-related studies (dotted curve), the general tendency over time is that there is an increasing proportion of glyphosate-related publications which satisfy the related search terms “safety” and/or “risk.” This brings us to conclude that safety and risk are relevant terms in present and recent research on glyphosate, as reflected by the indexed publications.
We have highlighted some of the studies which have been performed by chemical industry (A), the period of patent applications and first safety studies by independent researchers (B) and the time of introduction of GBH- tolerant transgenic crops (C), as this development has been identified as a most important single factor accelerating demand for GBH. Also, two important reviews (Giesy et al., 2000; Williams et al., 2000) were published around the time when several important national and international patents on glyphosates expired (D). The reviews are syntheses of evidence available at the time, notably including data and conclusions from numerous studies performed by industry. These industry reports had been reviewed by US authorities (EPA and FDA) but were recently found to lack the standards of peer reviewed studies (Samsel and Seneff, 2015b; Cuhra, 2015c). In addition, the actual reports from the laboratory work, specification of methodology, chemistry etc, have previously been inaccessible for independent verification, due to commercial interest. Several important studies were subsequently published by independent scientists (not affiliated with industry) presenting findings on higher toxicity in test animals and environment, and thus challenging the previously accepted view of negligible toxicity toward non-target organisms. Series of new findings also focused on effects in aquatic environments, finding evidence of higher toxicity toward amphibians (Relyea, 2005) and invertebrates. We have previously presented a small review of this evidence (Cuhra et al., 2013).
Resistant weeds and the lowered costs of GBH in recent years (after the patents expired) have led to crops being subjected to increasing application rates of glyphosates. Studies on glyphosate have investigated effects on soil microbiomes (Kremer and Means, 2009) and as an important parallel, on gastrointestinal communities in consumers (Samsel and Seneff, 2013). Also, a review of rodent studies and analysis of common commercial types of feed formulations for laboratory animals, have disclosed that such feed to a surprising extent is contaminated by various toxins and pesticide residues, including glyphosate (Mesnage et al., 2015c). These contaminations can affect the controls in the experiments and induce false positives as well as mask relevant effects. Thus, such feed-quality issues are a serious setback for the entire analytic community which depends on trustworthy data from rodent studies. The findings indicate fundamental systemic defects in rodent studies in general.
Historic Data on Glyphosate Non-Toxicity
For regulatory assessment of glyphosate health effects by US Food and Drug Administration (US-FDA) and parallel regulatory assessment of glyphosate ecotoxicity by US Environmental Protection Agency (US-EPA), the manufacturer carried out a wide range of laboratory testing in 1975–1985, in the first decade following its introduction. The tests were primarily performed by private subcontracting analytical laboratories according to established protocols at that time and submitted as evidence for regulatory assessment. Archive reports of numerous of these tests have been accessed but will not be discussed in detail here, other than noting that this archive base is not of peer-reviewed standard but rather has three levels of quality control; (a) at the performing laboratory, (b) at the commissioning industry, and (c) at the regulatory authority.
Through FOIA requests (FOIA, 2009, 2011) we have re-evaluated specific documentation extracted from the archives of the US Environmental Protection Agency (US EPA) and demonstrated faults in historical data assessing glyphosate toxicity in aquatic invertebrates. For example conclusions had been changed, regulatory importance exaggerated and wrong type of glyphosate had been tested (Cuhra, 2015c). Further investigations into industry safety-studies on glyphosate disclose notable early indications of carcinogenicity in rodents, albeit in high doses. A 2-year industry feeding study of glyphosate technical acid in rat showed significantly heightened incidences of tumors in high dose groups (US EPA, 1983). The industry applicant reported this to the US EPA including raw data and documentation. In a string of memos and letters these results were discussed internally in the EPA. Following (a) grouping of adenoma and carcinoma detections in treatment groups and in controls, and (b) re-evaluations of original histological slides from organs and tumor tissue, it was concluded that the incidence of tumors in treatment groups was not higher than controls (US EPA, 1983). Aspects of this case have recently been reviewed (Samsel and Seneff, 2015a) although the authors do not exhaustively discuss the role of the regulatory interpretation. A parallel case was highlighted in a 1985 internal EPA memorandum on false positives which points out methodological faults and mistakes in a 24-month study of glyphosate in mouse (unspecified type of glyphosate) submitted as evidence of safety to the EPA (US EPA, 1985). The main EPA argument against the interpretation presented by the applicant relates to the cancer incidence in the control group, which was claimed to be comparable to the treatment groups. The internal EPA memorandum explicitly states that the industry applicant interpretation should be overruled. The EPA conclusion from the statistical review is that the data demonstrates that glyphosate at 5000 and 30,000 ppm levels in feed, induce renal tubular adenomas.
Thus, we see buried historical evidence from two long-term studies of glyphosate in rodents, indicating carcinogenicity. The main reason these studies failed to achieve regulatory importance, seems to be the approval process conducted internally in the US EPA, which re-interpreted data and modified conclusions. It must be noted however, that both of these studies used high dosage of glyphosate and thus are not indications of tumorigenicity at lower dosage, such as found in a controversial recent two-year rat study with a glyphosate commercial formulation given in drinking water (Séralini et al., 2014).
GBH Aimed at New Targets
Agrochemicals such as GBHs will affect both the quality of agricultural products as well as the surrounding environment, notably as chemical residue levels in agricultural commodities and as impact on non-target organisms respectively.
Glyphosate was originally developed and patented as a broad-spectrum herbicide, with disrupting activity and lethal effects toward a broad spectrum of plants with active photosynthesis. Therefore, the use of glyphosate was restricted to pre-plant clearance of agriculture fields or forestry. In those early days of glyphosate use, the GBHs were not applied onto growing crops and thus the question of residues accumulating in plant material intended for consumption was less relevant. Furthermore, it was assumed that plants do not take up glyphosate from soil and thus even if a soil reservoir of un-metabolized glyphosate had been allowed to build up in agriculture soils, it was not perceived to be a problem. However, two developments in modern industrial agriculture have brought new challenges: (1) transgenic bypassing of the vital plant EPSPS (5-enolpyruvylshikimate-3-phosphate synthase) metabolic pathway, which is specifically targeted by glyphosate, allows for herbicide application onto growing crops, and (2) increasing use of GBHs as desiccants to kill and force-ripen semi-mature crops. Both of these developments have resulted in substantial amounts of GBHs being applied onto the crops intended for feed and food use. Thus, (1) the target organisms must be re-defined, and (2) the chemicals intended to eradicate weeds, have increasingly found a way into the food and feed supply of consumers.
In 2012 EFSA, the European Food Safety Authority, proposed a dramatic relaxation (increase) of maximum residue levels (MRLs) of herbicide glyphosate in lentils. The MRL for glyphosate in lentils was 0.1 mg/kg in the EU. The proposal aimed at raising the MRL for glyphosate in lentils to 15.0 mg/kg or alternatively 10.0 mg/kg, effectively by a factor of 150 (or 100) from the existing level. The proposal was submitted by the rapporteur member state Germany to the European Commission for approval on behalf of the applicant, Monsanto Europe (EFSA, 2012). The background for the proposal was findings of high residue levels of glyphosate in lentils grown in Canada. Residue levels ranging from 0.5 to 4.17 mg/kg were reported by the applicant, with one extreme high value of 8.8 mg/kg driving the proposed MRL target value of 15.0 mg/kg. At the same time when the application was submitted, a notification of food withdrawal from market was given by EU member state the Czech Republic, based on our detections of 10.5 mg/kg of glyphosate in lentils originating from Canada, which were confirmed by analysis of lentil samples taken from the Czech market (RASFF, 2012). This indicated that glyphosate residues in Canadian lentils occur at even more extreme values than envisaged in the requested relaxation of MRLs.
A communication from the Agriculture and Rural Development Department, Government of Alberta, Canada, describes the common practice of pre-harvest application of glyphosate to lentils in Canada as desiccant and recommends that the practice be terminated in harvest batches intended for export to the European Union (Agri-News, 2011). The newsletter discloses that glyphosate application immediately before harvest is widely used by farmers to force-ripen the lentil seed and though this practice is not estimated to conflict with the relatively relaxed MRLs in Canada, it will produce residues higher than the former MRLs for glyphosate in lentils in the European Union.
In the EU there seems to be lack of focus on the evidence of glyphosate use as desiccant and ripener in agriculture. Anecdotal evidence from rural areas in Denmark indicates that GBHs (Roundup) is routinely being used for ripening of wheat, and the practice is well-known from Germany; “ (…) in der EU seit einigen Jahren vermehrt Herbizide zur Sikkation von Erntebeständen, insbesondere von Getreide, Kartoffeln, Raps und Hülsenfrüchten, eingesetzt werden. Bei dieser Methode werden Herbizide kurz vor der Ernte direkt auf die zu erntenden Kulturpflanzen gespritzt. Das Totspritzen, wie die Sikkation treffender bezeichnet werden sollte, erleichtert durch gleichmäßig abgestorbene Pflanzen die Ernte (…)” (Brändli and Reinacher, 2012).
Further German studies (Haalck and Reinken, 2010) provide details on the practice of “Totspritzen” and document that a wide variety of herbicides in addition to glyphosates, such as glufosinate-ammonium, diquat, carfentzarone, cyanamid, cinidon-ethyl, and pyraflufen are used for this killing and forced ripening of crops.
The European Union maximum residue levels for glyphosate in barley grain are 20 ppm. For barley straw, the MRL is 200 ppm. These high MRLs are set to accommodate the use of glyphosate as desiccant in farming of barley. The main issue here may have implications far beyond the practicalities concerning the European Union maximum residue levels for glyphosate in lentils or barley. We find it disturbing that dominant agricultures are developing in such a way that toxins are used rather indiscriminately in order to ease harvesting. This use of herbicides is non-essential and from the perspective of both health of environment, hazardous. Again, here we see a development which contributes to the increasing total load of pesticides, and glyphosate in particular, into biota, fields and consumer organisms (Box 1).
Box 1. Glyphosate Facts
Facts 1: Global Omnipresence
• Glyphosate herbicides (GBHs) such as Roundup have been on the market since 1975 and their use is still increasing, making GBHs the primary category of pesticides world-wide. By volume and revenue, GBHs are globally dominant.
• Glyphosate is detected in water, air, animal feed, animal urine, and animal flesh. Glyphosate is also found in human food, human milk, and human urine.
Facts 2: Higher dosage and increased ingestion
• Regulation of glyphosate has gradually been relaxed, allowing for increasing maximum residue limits in important food and feed commodities.
• GBHs are used for late-season application and pre-harvest desiccation. Such practices cause high residue levels.
• Animal- and human consumer ingestion is increasing due to higher residue levels in food and increasing number of glyphosate sources.
Facts 3: Safety-assessements are flawed
• Reviews of older safety assessment studies of glyphosate have uncovered flaws and misinterpretations in the regulatory base.
• Numerous safety assessments have been performed with glyphosate technical acid instead of the glyphosate salts actually used in GBH herbicide formulations.
• Lack of labeling and low traceability of food/feed, combined with unknown levels of glyphosate in such biomass, is prohibitive for research on effects in consumers.
Facts 4: Recent developments
• Recently, regulators such as the EFSA have reduced the annual frequency of analysis for glyphosate residues in food and feed, giving glyphosate lower priority
• New research indicates that glyphosate should be recognized as having potentially more complex and severe effects on health and environment than previously assumed.
• Other research upholds that since humans and animal consumers do not have the EPSPS photosynthesis pathway, they will not be affected by glyphosate.
Residues in Plants and Food/Feed Products
Recognizing the fact that consumers are ingesting more glyphosate residues via our food supply, it is also relevant to review this exposure. Bio-active herbicides interact with biomass and ultimately get into soil and water systems through processes such as drifting, leaching, and surface runoff (Mensah et al., 2012). Glyphosate is present in ground water, human and animal urine, human breast milk, and farmed-animal flesh (Borggaard and Gimsing, 2008; Krüger et al., 2013, 2014; Honeycutt and Rowlands, 2014; Niemann et al., 2015). Thus, potential interaction with other stressors in biological systems or in the environment need to be studied in more realistic settings (Then, 2009; Nørgaard and Cedergreen, 2010; Bjergager et al., 2011). Glyphosate or GBH should not be evaluated or discussed in isolation. In organisms and biota exposure to glyphosate will co-occur with exposure to other pesticides. Monitoring programmes generally detect more than 7-8 different pesticides in single samples from the environment, and cocktails of multiple pesticides are routinely present in foods and feedstuffs (EFSA, 2014). In spite of that, current testing regimes for relevant agrochemicals are predominately based on acute exposure (short term) and specific testing of isolated single chemicals (Martin et al., 2003; Nørgaard and Cedergreen, 2010). Studies of combinatorial effects of multiple toxins are however increasingly acknowledged as missing (van Haver et al., 2008; Al-Gubory, 2014).
In the late 1997–1999, levels of 1.9–4.4 mg/kg glyphosate was found in Roundup Ready soy plant parts other than the grains, and 0.1–1.8 mg/kg was found in the grains (Arregui et al., 2004). A study from the US noted that repeated herbicide applications increased the residue levels of both glyphosate and AMPA in the soybeans. At three applications the highest residue level found was at 3.08 mg/kg for glyphosate and 25 mg/kg for AMPA (Duke et al., 2003). Thus, applications closer to time of harvest induce relatively high residue levels in the soybeans, leading to high residues in commodities.
The scarcity of published data on glyphosate residues in glyphosate-tolerant crops such as Roundup-ready soybean is unfortunate. In this situation estimates must be based on the few existing data: an earlier publication from Duke et al. (2003), our recent data from USA (Bøhn et al., 2014) and Argentina (Then, 2013). Data presented in Cuhra (2015b, Figure 1) are recalculated from Bøhn et al. (2014) and shows AMPA as glyphosate equivalents conforming to the FAO standards of the data presented by Then (2013). Average glyphosate-equivalent residue concentrations are 11.87 mg/kg in tests of soybeans from USA and 39.87 mg/kg in tests of soybeans from Argentina. These average concentrations are in compliance with the maximum residue levels defined by the US FDA (40 mg/kg) and the results from USA are also in compliance with the EU MRL of 20 mg/kg. However, individual samples from Argentina exceed current MRLs.
In comparison to the level of glyphosate in crops, other pesticides are typically found in much lower concentrations. For example, in US soybeans we found Fluazifop-P (0.078 mg/kg, one sample “Roundup-ready”), malathion (0.02 mg/kg, one sample “conventional”), and dieldrin (0.002 mg/kg, one sample, “organic”). In pooled samples alpha-endosulfane, trans-nonachlor, and trans-chlordane was found at levels close the detection limit of 0.05 μg/kg (Bøhn et al., 2014).
Thus, there are striking concentration differences between glyphosate and other pesticides in food and feed crops. Contrary to other pesticides that are measured in low ppm or ppb levels, glyphosate is detected at ppm-levels, orders of magnitude higher.
Given the very large quantities of soybean material produced, it is relevant to calculate or estimate the total amount of residues thus transported and mediated to consumers (mainly farm animals). The majority (82%) of global soybean production stems from glyphosate tolerant soy (James, 2014). The total global production in the 2013/14 growing season, was estimated to be 320 million ton (USDA, 2016), of this 290 million ton is estimated to be cultivated in glyphosate-tolerant varieties (Roundup ready soy). Based on the findings of residue concentrations in US soybeans (11.87 mg/kg glyphosate-equivalents) the quantity of glyphosate residues which are accumulated, translocated, and consumed via glyphosate tolerant soy, is ~3440 ton. Recalculating by using the data from Argentina (39.87 mg/kg glyphosate-equivalents), this figure could be as high as 11560 ton (Cuhra, 2015a). However large it may seem, this quantity is just a fraction of the total load of glyphosate herbicides applied in soybean cultivation. Exact figures are difficult to obtain, not least since the cultivation of transgenic soybean is continuously expanding and application rates of glyphosate active ingredient are increasing. Based on data from the US Soybean association, the USDA, the Penn State University online Agronomy-Guide and similar readily accessible sources it is not unreasonable to use production figures of 2.5–2.9 ton/ha for present soybean yield and estimates of 90 Million hectares for the total area currently in global cultivation with glyphosate tolerant soybean. Based on the same sources, realistic seasonal application rates for glyphosate herbicides are likely not < 1.7 kg active ingredient per hectare, probably closer to 2.5 kg. A conservative estimate can be based on the USDA maximal single-pass application rate of 1.5 lb/acre, and total area in cultivation. With one seasonal pass of maximum allowed application, the total quantity of glyphosate active ingredient applied on glyphosate tolerant soybean globally would be in the magnitude of 153,000 ton. This would indicate that roughly 14% of the global production of glyphosate is used in agriculture of glyphosate-tolerant soybean. Assessing the application figures via the available production data for soybean yield, the estimates are similar, ~140,000 ton. These figures indicate that 2–7% of the applied glyphosate active ingredient is accumulated in the soybean commodity. This represents a sizeable amount of pesticide residues directed at consumers, via the herbicide tolerant GM crops. We argue that regulators/governments need to respond and re-evaluate the potential human and animal health risks from this exposure.
Duke et al. (2003) found a low glyphosate/AMPA ratio in soybean following late application. The data on residues in Roundup ready soybean from Iowa show a similar trend (Bøhn et al., 2014). However, the glyphosate/AMPA ratios in analyzed samples were found to be inconsistent. Glyphosate is known to interact with biochemical processes in metabolism of transgenic glyphosate tolerant plants (Zobiole et al., 2011). The scarcity of published data on glyphosate residues accentuates the relevance of further investigating the dynamics of glyphosate degradation and transformation in plants. EFSA noted in the annual monitoring report (EFSA, 2015b) that for certain pesticides covered by the 2013 European coordinated monitoring programme (EUCP), including glyphosate, the number of determinations reported was significantly below the number needed to derive statistically sound conclusions. In comparison to e.g., some pyrethroids or organophosphates, the number of analyzed samples for glyphosate was ~25 times lower (chlorpyriphos–70943 samples, glyphosate–2866 samples). The reason is that glyphosate is impossible to include in multi-residual methods as it requires the application of a single and specific method, which is expensive, demanding, and time consuming. Only a limited number of laboratories are able to perform it. For the same reason, not only the number of samples, but also the number of commodities involved in monitoring programmes (and thus also in risk assessment) are limited. Analyses were performed on a limited set of commodities (e.g., apples; oats; rye; wine, grapes, wheat) in the EU monitoring programme in 2013. In spite of this, a total of 7.9% of samples were glyphosate positive (i.e., above LOQ). In some commodities, high ratio of glyphosate positive samples were found—e.g., for oats, 44% of samples were found as positive. According to the EFSA, reporting countries should extend the scope of the analytical methods used for enforcement of MRLs to make sure that the detection rate and the MRL exceedance rate is not biased by the low number of determinations or lack of data from certain countries. It is clear, that at present there is lack of reliable and representative results for most of food commodities in the food basket. In addition, the main metabolite AMPA is not included in coordinated EU monitoring programme.
Accepted Levels of Glyphosate Residues
Regulatory threshold of accepted levels of glyphosate residues are continuously being raised. At present the maximal residue levels (MRLs) of glyphosate in soybean in the USA has been increased from 20 up to 40 mg/kg in the fall of 2013. Again, we accentuate that such ppm-levels are high when compared to other pesticide active ingredients such high MRLs should only be accepted for compounds with very low toxicity. Review of regulatory documents such as the US EPA (1993) RED on glyphosate shows that such MRLs are defined pragmatically; to accommodate existing residue levels and existing agriculture practice (US EPA, 1993). Furthermore, we find that even the recently raised acceptance levels will not be enough for the concentrations of residues found in the transgenic soybean material tested in Argentina.
The global annual soy production equals ~43 kg per capita. Of this quantum, ~39 kg is from glyphosate tolerant varieties. Direct human consumption of soy is minimal as the majority of the global production at present is utilized in production of feed for farmed animals. Many species of farmed animals (cattle, poultry, pigs, fish, prawns etc) are fed diets with a considerable proportion of soy. Such feeding is daily and throughout the whole life span. This fact alone accentuates the relevance of adequate testing for chronic exposure to, and potential effects from, glyphosate residues. A recent report of glyphosate residues in aborted and malformed piglets from sows in intensive animal farming is remarkable albeit inconclusive (Krüger et al., 2014). Although this important indication necessitates further research, we note that due to faults in methodology, lack of a proper control group, and missing information on feed composition, the reported abortion rates and malformations cannot be irrefutably linked to lifelong feeding with GMO ingredients containing normal levels of glyphosate residues.
Environmental Impact Quotient (EIQ) of Glyphosates
Herbicides and other chemical substances intended for dispersal into the environment are evaluated for unintended and undesirable effects in indicator organisms representing non-target species. The results of testing is extrapolated to other taxonomic groups and extended to ecosystem levels, thus providing information for regulatory decisions. Furthermore, indicators from e.g., oral ingestion in representative test species, dermal exposure, inhalation, and cell culture studies are amongst the indicators important for assessment of effects on human biology. Kovach et al. (1992) established the Environmental Impact Quotient for pesticides (EIQ) as a measure to condense into one indicative denominator the relative toxicity of specific chemical compounds, by collecting fragmented evidence on effects in a variety of indicators. Main components of the EIQ are three categories of effects defined as; Farmworker component, Consumer component, and Ecological component. Such relative indicators can be used as general comparators and the process involved in determining the EIQ of a novel pesticide compound can in itself be a useful exercise for regulators and stakeholders. However, the validity of such a relative indicator is dependent on regular revisions of the basis, the scientific evidence, which supplies numerical values to the individual factors in the equation from which the quotient is calculated.
The Dynamics of Pesticide Regulation
In a time with considerable confusion regarding possible toxic effects of glyphosate herbicide toward health and environment, with contradictory findings on potential impacts and strong voices arguing on one side for precaution, on the other side incentives for continued high volume use of a chemical, we find it useful to mention the key elements which constitute the basis for regulation. In a commercially driven market economy, the dominating societal model in the world, industry interests seek to market and employ products. Some products may have unwanted consequences. In general, the regulation of potentially toxic chemicals, e.g., pesticides, is largely based on scientific information produced by the industry which often has strong financial incitement for unrestricted use. Thus, in society there is an antagonistic tension between commercial vs. public interests concerning the regulation of global and local application of e.g., glyphosate herbicides. This leads to a dynamic interplay driven by two main vectors, of which one represents commercial forces (in this case primarily manufacturing chemical industry and farmers), and the other represents societal interest (health, environmental protection, qualitative requirements) (Cuhra, 2015c).
The arguments supporting and enhancing the opposing vectors, are furnished by scientists and other professionals working within private sector research firms, in publicly funded university laboratories, in regulatory authorities, as consultants or in non-governmental organizations representing defined interests. All of these, we commonly call “experts.” Resulting policy should be a careful balance of these expert opinions, based on factual findings from e.g., laboratory testing (Figure 2). Thus, when scientific evidence shows that a compound or groups of compounds has low toxicity for consumers and environment, restrictions on use are relaxed (society accepts more). However, in the opposite case, if science demonstrates that compounds are more toxic than previously assumed, their penetration into environment and food chains should be reduced through regulatory measures (society accepts less,—such as in the cases of DDT and PCBs).
 Figure 2. Conceptual model of glyphosate toxicity and maximum residue level (MRL) over time. The paradox of glyphosate regulation. Red dots symbolize evidence of toxic effects, measured as a relative denominator defined in the left y-axis “Glyphosate toxicity.” The red arrow shows the trend over time, as more evidence demonstrates higher toxicity. Green dots symbolize acceptance (MRLs) on a relative scale (the right side y-axis). Green arrow shows trend over time, as MRLs are increased. In science-based policy evidence of higher toxicity should lead to lowering of acceptance levels (MRLs). In the case of glyphosate, the development is the opposite: increasing acceptance is positively correlated with toxicity.
Figure 2. Conceptual model of glyphosate toxicity and maximum residue level (MRL) over time. The paradox of glyphosate regulation. Red dots symbolize evidence of toxic effects, measured as a relative denominator defined in the left y-axis “Glyphosate toxicity.” The red arrow shows the trend over time, as more evidence demonstrates higher toxicity. Green dots symbolize acceptance (MRLs) on a relative scale (the right side y-axis). Green arrow shows trend over time, as MRLs are increased. In science-based policy evidence of higher toxicity should lead to lowering of acceptance levels (MRLs). In the case of glyphosate, the development is the opposite: increasing acceptance is positively correlated with toxicity.In a previous commentary, we have reflected upon the quality of evidence supporting the notion of glyphosate non-toxicity, finding that serious flaws confuse the current regulatory basis (Cuhra, 2015c).
Socioeconomic Aspects
Important societal challenges related to production of glyphosate-tolerant crops such as Roundup-ready soybean include ecological damage through deforestation and degradation of natural habitats (Pengue, 2005) and glyphosate pollution of the environment. The large-scale cultivation of glyphosate-tolerant crops, such as Roundup-ready soy (RR-soy), RR-maize, and RR-canola has also been identified as a main cause for emergence and widespread occurrence of numerous glyphosate-resistant agricultural weeds (Duke and Powles, 2008; Benbrook, 2012). The weed-challenges will be met with alternative and more potent mixes of herbicides (Green, 2009), whereby older and arguably more toxic herbicides, such as atrazine, may be reintroduced (Binimelis et al., 2009). This development has been linked to increased occurrence of severe medical problems in farmers and farm village populations in Argentina, in areas where Roundup-ready soybean is produced (Vazquez and Nota, 2011).
Here, the evolution of glyphosate use and risk-assessment has been defined as five distinct periods (each a decade) following the discovery and commercialization of glyphosate in 1975 (Figure 3). The first decade (1975–1985) represents “glyphosate optimism.” Glyphosate was discovered as a very efficient herbicide, with a systemic action on a broad spectrum of agriculture weeds. At that time, glyphosate was perceived to have very low toxicity toward users, non-target organisms and consumers of agriculture produce. The following two decades (1986–2005) saw global implementation of glyphosate based herbicides such as Roundup and a dramatic increase in glyphosate use. The introduction and successful commercialization of several glyphosate-tolerant genetically modified crops in 1995 was a development later identified as the most important factor accelerating the use of glyphosate herbicides (Charles, 2001; Duke and Powles, 2008; Benbrook, 2012, 2016). However, although the use of glyphosate has accelerated even further in the following decade (2006–2015) this has also been a decade of increasing and sobering challenges, notably caused by the advent of tolerant and resistant weed species, globally disrupting the efficacy of this agrochemical system. We define this latest decade “the decade of glyphosate skepticism” in our model. Numerous research programs, reviews and laboratory findings have documented that the safety assumptions of glyphosate are mature for revision. The decade culminated with a string of published evidence in 2015 detailing the challenging issues (Mesnage et al., 2015a; Samsel and Seneff, 2015b) even concluding that glyphosate should be categorized as a probable carcinogen (Guyton et al., 2015), in contrast to previously accepted conclusions concerning these chemicals. EFSA recently reviewed the evidence of glyphosate carcinogenicity and concluded that glyphosate is not a carcinogen (EFSA, 2015c). Other research in 2015 indicated that previous assumptions of safety, have in part been based on flawed evidence or misinterpretations (Cuhra, 2015c; Samsel and Seneff, 2015a).
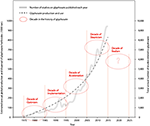 Figure 3. Concept model to visualize how societal perception of glyphosate has evolved through five decades (1975–2025), related to trends in glyphosate use (Benbrook, 2016) and increasing annual rate of publications on glyphosate.
Figure 3. Concept model to visualize how societal perception of glyphosate has evolved through five decades (1975–2025), related to trends in glyphosate use (Benbrook, 2016) and increasing annual rate of publications on glyphosate.Future Developments
Agricultural industry in general depends on more-or-less toxic pesticides. This is a generally accepted normality for conventional agriculture, which has developed gradually since the latest great war (Alston et al., 2010) and now constitutes an “agroecological-prison-situation,” in which pesticides and other chemicals are now unavoidable in order to make industrial farming cost-effective. Thus, farmers are trapped and dependent on a combination of selected seeds and selected poisons.
Despite the challenges associated with both the continued use of glyphosate as the principal herbicide and the continued cultivation of glyphosate tolerant crops, there are few attractive chemical-biotechnological alternatives at present. Several crop varieties tolerant to herbicidal chemicals glufosinate-ammonium, dicamba, and 2,4-D are currently either in development, awaiting approval or already on the market. But, it is still an unresolved issue whether these crop varieties and agrochemical systems (which are relying on “old” herbicide technology) are as efficient, cost-effective or “better or worse” for the receiving environment, as the existing glyphosate-tolerant varieties currently available. Despite the aforementioned challenges posed by glyphosate-tolerant GMOs, several large biotech firms are now releasing “second-generation” glyphosate-tolerant cultivars touted as being even more efficient. Developing a new herbicide and getting it approved for use is very costly. According to some estimates, the financial investments of industry can amount to US $180 million and the regulatory approval can take a decade (Smith et al., 2008; McDougall, 2010). Furthermore, it is challenging for industry to meet societal demands in such developments; new compounds are expected to have high target specificity and low general toxicity (for the environment, the users and the consumers of agricultural commodities). The biotech-agrochemical industry therefore adheres to two general strategies: it develops and registers new transgenic cultivars and chemical compounds for the market (ISAAA, 2014); and it uses existing chemical compounds in new ways, notably through introduction of transgenic varieties that tolerate higher doses of approved agrochemicals such as glyphosate (e. g., Cao et al., 2012, 2013). The role of glyphosate herbicides can therefore be expected to remain predominant in global industrial agriculture, especially in cultivation of glyphosate-tolerant varieties. As such, it is relevant to consider the possible benefits vs. challenges associated with continued or increased glyphosate use.
Returning to the history of glyphosate as depicted in Figure 3, we suggest that the decade which we are entering at the time of this writing, should be later seen as the period of “glyphosate realism.” Hopefully a time when glyphosate will be recognized as a chemical which has to be stewarded carefully and restricted. This would allow that glyphosate can be used sensibly, in moderation, and play a reduced role in global agriculture as the lesser evil, until an alternative is found.
Returning to Shakespeare, let us join the young prince of Denmark as he exclaims to his friends: “Why, then,’tis none to you, for there is nothing either good or bad, but thinking makes it so. To me it is a prison” (Hamlet, Act 2, scene 2).
Conclusion
The recognized higher toxicity and the stronger potential for negative effects on health and environment should be important arguments for restrictions in use of glyphposate and GBHs. Despite this evidence, regulatory authorities have gradually allowed more sources of glyphosate into the food-supply and higher residue levels, in an ongoing development contrary to toxicological principles and common sense.
Author Contributions
All listed authors have contributed to the work. The corresponding author initiated and structured the manuscript, drew the figures, coordinated various input and elaborated the wording.
Funding
The research was funded by the Research Council of Norway through NFR Project 184107/S30 LAND: A new model approach to assess genetically modified plants: their ecotoxicity and potential interactions with environmental pollutants.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Agri-News (2011). Lentil Producers Unable to Use Glyphosate for Pre-harvest Weed Control. Edmonton: Newsletter 15/08/2011 Agriculture and Rural Development Department, Government of Alberta.
Al-Gubory, K. H. (2014). Environmental pollutants and lifestyle factors induce oxidative stress and poor prenatal development. Reprod. Biomed. Online 29, 17–31. doi: 10.1016/j.rbmo.2014.03.002
Alston, J. M., Beddow, J. M., and Pardey, P. G. (2010). “Global patterns of crop yields and other partial productivity measures and Prices,” in The Shifting Patterns of Agricultural Production and Productivity Worldwide, eds J. M. Alston, B. A. Babcock, and P. G. Pardey (Ames, IA: The Midwest Agribusiness Trade Research and Information Center; Iowa State University), 39–61.
Amrhein, N., Deus, B., Gehrke, P., and Steinrücken, H. C. (1980). The site of the inhibition of the shikimate pathway by Glyphosate. Plant Phys. 66, 830–834. doi: 10.1104/pp.66.5.830
Arregui, M. C., Lenardon, A., Sanchez, D., Maitre, M. I., Scotta, R., and Enrique, S. (2004). Monitoring glyphosate residues in transgenic glyphosate-resistant soybean. Pest Manag. Sci. 60, 163–166. doi: 10.1002/ps.775
Benbrook, C. M. (2012). Impacts of genetically engineered crops on pesticide use in the U.S. – the first sixteen years. Environ. Sci. Eur. 24, 1–13. doi: 10.1186/2190-4715-24-24
Benbrook, C. M. (2016). Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 28, 1–15. doi: 10.1186/s12302-016-0070-0
Binimelis, R., Pengue, W., and Monterroso, I. (2009). “Transgenic treadmill”: responses to the emergence and spread of glyphosate-resistant johnsongrass in Argentina. Geoforum 40, 623–633. doi: 10.1016/j.geoforum.2009.03.009
Bjergager, M. B. A., Hanson, M. L., Lissemore, L., Henriquez, N., Solomon, K. R., and Cedergreen, N. (2011). Synergy in microcosms with environmentally realistic concentrations of prochloraz and esfenvalerate. Aquat. Toxicol. 101, 412–422. doi: 10.1016/j.aquatox.2010.11.004
Bøhn, T., Cuhra, M., Traavik, T., and Fagan, J. (2016). “Are ready for market genetically modified, conventional and organic soybeans substantially equivalent as food and feed?,” in Genetically Modified Organisms in Food, eds R. R. Watson and V. R. Preedy (Amsterdam: Elsevier), 181–191.
Bøhn, T., Cuhra, M., Traavik, T., Sanden, M., Fagan, J., and Primicerio, R. (2014). Compositional differences in soybeans on the market: Glyphosate accumulates in Roundup Ready GM soybeans. Food Chem. 153, 207–215. doi: 10.1016/j.foodchem.2013.12.054
Bonny, S. (2011). Herbicide-tolerant Transgenic Soybean over 15 years of cultivation: pesticide use, weed resistance, and some economic issues. The case of the USA. Sustainability 3, 1302–1322. doi: 10.3390/su3091302
Bonny, S. (2014). Taking stock of the genetically modified seed sector worldwide: market, stakeholders, and prices. Food Secur. 6, 525–540. doi: 10.1007/s12571-014-0357-1
Borggaard, O. K., and Gimsing, A. L. (2008). Fate of glyphosate in soil and the possibility of leaching to ground and surface waters: a review. Pest Manag. Sci. 64, 441–456. doi: 10.1002/ps.1512
Cao, G., Liu, Y., Liu, G., Wang, J., and Wang, G. (2012). A Novel 5-Enolpyruvylshikimate-3-phosphate synthase shows high glyphosate tolerance in Escherichia coli and tobacco plants. PLoS ONE 7:e38718. doi: 10.1371/journal.pone.0038718
Cao, G., Liu, Y., Liu, G., Wang, J., and Wang, G. (2013). Draft genome sequence of pseudomonas strain P818, isolated from glyphosate-polluted soil. Genome Announc. 1, e01079–e01013. doi: 10.1128/genomea.01079-13
Cattani, D., de Liz Oliveira Cavalli, V. L., Heinz Rieg, C. E., Domingues, J. T., Dal-Cim, T., Tasca, C. I., et al. (2014). Mechanisms underlying the neurotoxicity induced by glyphosate-based herbicide in immature rat hippocampus: Involvement of glutamate excitotoxicity. Toxicology 320, 34–45. doi: 10.1016/j.tox.2014.03.001
Cavalli, V. L. D. O., Cattani, D., Rieg, C. E. H., Pierozan, P., Zanatta, L., Parisotto, E. B., et al. (2013). Roundup disrupts male reproductive functions by triggering calcium-mediated cell death in rat testis and Sertoli cells. Free Radic. Biol. Med. 65, 335–346 doi: 10.1016/j.freeradbiomed.2013.06.043
Cuhra, M. (2015a). Ecotoxicological assessment of Roundup-ready soybean agriculture investigated in a D. magna Model. Tromsø: Dissertation for the degree of Philosophiae Doctor. Faculty of Health Sciences, University of Tromsø Norway.
Cuhra, M. (2015b). Review of GMO safety assessment studies: glyphosate residues in Roundup Ready crops is an ignored issue. Environ. Sci. Eur. 27, 1–14. doi: 10.1186/s12302-015-0052-7
Cuhra, M. (2015c). Glyphosate nontoxicity: the genesis of a scientific fact. J. Biol. Phys. Chem. 15, 89–96. doi: 10.4024/08CU15A.jbpc.15.03
Cuhra, M., Traavik, T., and Bøhn, T. (2013). Clone-and age-dependent toxicity of a glyphosate commercial formulation and its active ingredient in Daphnia magna. Ecotoxicology 22, 251–262. doi: 10.1007/s10646-012-1021-1
Cuhra, M., Traavik, T., and Bøhn, T. (2014). Life cycle fitness differences in Daphnia magna fed Roundup-Ready soybean or conventional soybean or organic soybean. Aquaculture Nutr. 21, 702–713. doi: 10.1111/anu.12199
Cuhra, M., Traavik, T., Dando, M., Primicerio, R., Holderbaum, D. F., and Bøhn, T. (2015). Glyphosate-residues in roundup-ready soybean impair Daphnia magna life-cycle. J. Agric. Chem. Environ. 4, 24. doi: 10.4236/jacen.2015.41003
Defarge, N., Takács, E., Lozano, V. L., Mesnage, R., Vendômois, J. S., Seralini, G. E., et al. (2016). Co-formulants in glyphosate-based herbicides disrupt aromatase activity in human cells below toxic levels. Int. J. Environ. Res. Public Health 13, 264. doi: 10.3390/ijerph13030264
Duke, S. O., and Powles, S. B. (2008). Glyphosate: a once-in-a-century herbicide. Pest Manag. Sci. 64, 319–325. doi: 10.1002/ps.1518
Duke, S. O., Lydon, J., Koskinen, W. C., Moorman, T. B., Chaney, R. L., and Hammerschmidt, R. (2012). Glyphosate effects on plant mineral nutrition, crop rhizosphere microbiota, and plant disease in glyphosate-resistant crops. J. Agric. Food Chem. 60, 10375–10397. doi: 10.1021/jf302436u
Duke, S. O., Rimando, A. M., Pace, P. F., Reddy, K. N., and Smeda, R. J. (2003). Isoflavone, glyphosate, and aminomethylphosphonic acid levels in seeds of glyphosate-treated, glyphosate-resistant soybean. J. Agric. Food Chem. 51, 340–344. doi: 10.1021/jf025908i
EFSA (2012). European Food Safety Authority. Reasoned opinion – Modification of the existing MRL for glyphosate in lentils. EFSA J. 10:2550. doi: 10.2903/j.efsa.2012.2550
EFSA (2014). The 2012 European Union Report on pesticide residues in food. EFSA J. 12:3942. doi: 10.2903/j.efsa.2014.3942
EFSA (2015a). Renewal Assessment Report – Glyphosate – Report and Proposed Decision (18 December 2013, Revised 29 January 2015, 31 March 2015). Parma: European Food Safety Authority (EFSA).
EFSA (2015b). SCIENTIFIC REPORT OF EFSA The 2013 European Union Report on Pesticide Residues in Food. Parma: European Food Safety Authority (EFSA).
EFSA (2015c). Glyphosate: EFSA Updates Toxicological Profile – Glyphosate is not a Cancerogen. (12 November 2015) Available online at: www.efsa.europa.eu/en/press/news/151112
FOIA (2009). The Freedom of Information Act. The National security Archive. Available online at: www.gwu.edu/~nsarchiv/nsa/foia.html
FOIA (2011). 2010-2011 Agency FOIA Success Stories: Creating a Culture of Transparency. Available online at: www.foia.gov/iso/opa/foia/oip-1103111.html
Folmar, L. C., Sanders, H. O., and Julin, A. M. (1979). Toxicity of the herbicide glyphosate and several of its formulations to fish and aquatic invertebrates. Arch. Environ. Contam. Toxicol. 8, 269–278. doi: 10.1007/BF01056243
Gasnier, C., Dumont, C., Benachour, N., Clair, E., Chagnon, M. C., and Séralini, G. E. (2009). Glyphosate-based herbicides are toxic and endocrine disruptors in human cell lines. Toxicology 262, 184–191. doi: 10.1016/j.tox.2009.06.006
Giesy, J., Dobson, S., and Solomon, K. R. (2000). Ecotoxicological risk assessment for Roundup herbicide. Rev. Environ. Contam. Toxicol. 167, 35–120. doi: 10.1007/978-1-4612-1156-3_2
Grandjean, P., and Landrigan, P. J. (2014). Neurobehavioural effects of developmental toxicity. Lancet Neurol. 13, 330–338. doi: 10.1016/S1474-4422(13)70278-3
Green, J. M. (2009). Evolution of glyphosate-resistant crop technology. Weed Sci. 57, 108–117. doi: 10.1614/WS-08-030.1
Gress, S., Lemoine, S., Seralini, G. E., and Puddu, P. E. (2015). Glyphosate-based herbicides potently affect cardiovascular system in mammals: review of the literature. Cardiovasc. Toxicol. 15, 117–126. doi: 10.1007/s12012-014-9282-y
Guyton, K. Z., Loomis, D., Grosse, Y., El Ghissassi, F., Benbrahim-Tallaa, L., Guha, N., et al. (2015). Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Lancet Oncol. 16 490–491. doi: 10.1016/S1470-2045(15)70134-8
Haalck, F., and Reinken, B. (2010). Sikkation, ein Grund zum Fragen. Available online at: http://www.schattenblick.de/infopool/umwelt/redakt/umko0005.html
Heap, I. (2015). The International Survey of Herbicide Resistant Weeds. Available online at: http://weedscience.org/
Honeycutt, Z., and Rowlands, H. (2014). Glyphosate Testing Report: Findings in American Mothers’ Breast Milk, Urine and Water. Moms Across America. Available online at: http://www.momsacrossamerica.com/glyphosate_testing_results
Howe, C. M., Berrill, M., Pauli, B. D., Helbing, C. C., Werry, K., and Veldhoen, N. (2004). Toxicity of glyphosate-based pesticides to four North American frog species. Environ. Toxicol. Chem. 23, 1928–1938. doi: 10.1897/03-71
ISAAA (2014). GM Approval Database. International Service for the Acquisition of Agri-biotech Applications (ISAAA). Available online at: http://www.isaaa.org/gmapprovaldatabase/
Kovach, J., Tette, J., Degni, J., and Petzoldt, C. (1992). A method to measure the environmental impact of pesticides. New Yorks Food Life Sci. Bull. 139, 1–8.
Kremer, R. J., and Means, N. E. (2009). Glyphosate and glyphosate-resistant crop interactions with rhizosphere microorganisms. Eur. J. Agron. 31, 153–161. doi: 10.1016/j.eja.2009.06.004
Krüger, M., Schrodl, W., Neuhaus, J., and Shehata, A. A. (2013). Field investigations of glyphosate in urine of Danish dairy cows. J. Environ. Anal. Toxicol. 3:100186. doi: 10.4172/2161-0525.1000186
Krüger, M., Schrödl, W., Pedersen, I., and Shehata, A. A. (2014). Detection of glyphosate in malformed piglets. J. Environ. Anal. Toxicol. 4, 1–2. doi: 10.4172/2161-0525.1000230
Malhotra, R. C., Ghia, D. K., Cordato, D. J., and Beran, R. G. (2010). Glyphosate-surfactant herbicide-induced reversible encephalopathy. J. Clin. Neurosci. 17, 1472–1473. doi: 10.1016/j.jocn.2010.02.026
Martin, J. D., Crawford, C. G., and Larson, S. J. (2003). Pesticides in Streams. National Water Assessment Program (NAWQA). Available online at: http://water.usgs.gov/nawqa
McDougall, P. (2010). The Cost of New Agrochemical Product Discovery, Development and Registration in 1995, 2000 and 2005-8. Report for European Crop Protection association and Crop Life America. Vineyard Business Centre, Midlothian.
Mensah, P. K., Muller, W. J., and Palmer, C. G. (2012). Using growth measures in the freshwater shrimp Caridina nilotica as biomarkers of Roundup (R) pollution of South African freshwater systems. Phys. Chem. Earth 50–52, 262–268. doi: 10.1016/j.pce.2012.08.003
Mesnage, R., Arno, M., Costanzo, M., Malatesta, M., Séralini, G. E., and Antoniou, M. N. (2015b). Transcriptome profile analysis reflects rat liver and kidney damage following chronic ultra-low dose Roundup exposure. Environ. Health 14, 70. doi: 10.1186/s12940-015-0056-1
Mesnage, R., Defarge, N., Rocque, L. M., de Vendômois, J. S., and Séralini, G. E. (2015c). Laboratory rodent diets contain toxic levels of environmental contaminants: implications for regulatory tests. PLoS ONE 10:e0128429. doi: 10.1371/journal.pone.0128429
Mesnage, R., Defarge, N., Spiroux de VendÓmois, J., and Séralini, G. E. (2015a). Potential toxic effects of glyphosate and its commercial formulations below regulatory limits. Food Chem. Toxicol. 84, 133–153. doi: 10.1016/j.fct.2015.08.012
Mesnage, R., Defarge, N., Vendômois, J. S., and Séralini, G. E. (2014). Major pesticides are more toxic to human cells than their declared active principles. BioMed Res. Int. 2014:179691. doi: 10.1155/2014/179691
Millstone, E., Brunner, E., and Mayer, S. (1999). Beyond substantial equivalence. Nature 401, 525–526. doi: 10.1038/44006
Niemann, L., Sieke, C., Pfeil, R., and Solecki, R. (2015). A critical review of glyphosate findings in human urine samples and comparison with the exposure of operators and consumers. J. Verbraucherschutz Lebensmittelsicherheit 10, 3–12. doi: 10.1007/s00003-014-0927-3
Nørgaard, K. B., and Cedergreen, N. (2010). Pesticide cocktails can interact synergistically on aquatic crustaceans. Environ. Sci. Poll. Res. 17, 957–967. doi: 10.1007/s11356-009-0284-4
Peixoto, F. (2005). Comparative effects of the Roundup and glyphosate on mitochondrial oxidative phosphorylation. Chemosphere 61, 1115–1122. doi: 10.1016/j.chemosphere.2005.03.044
Pengue, W. (2005). Transgenic crops in Argentina: the ecological and social Debt. Bull. Sci. Technol. Soc. 25, 314–322. doi: 10.1177/0270467605277290
RASFF (2012). EU Rapid Alert System for Food and Feed (RASFF) search results: Notification detail 2012.0172 dated 03/02/2012. Prague: Czech Republic Food Inspection.
Relyea, R. A. (2005). The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities. Ecol. Appl. 15, 618–627. doi: 10.1890/03-5342
Robin, M. M. (2008). Le Monde selon Monsanto : De la dioxine aux OGM, une multinationale qui vous veut du bien, ARTE France Développement. Paris: EAN 3453277601642.
Samsel, A., and Seneff, S. (2013). Glyphosate’s suppression of cytochrome P450 enzymes and amino acid biosynthesis by the gut microbiome: pathways to modern diseases. Entropy 15, 1416–1463. doi: 10.3390/e15041416
Samsel, A., and Senefff, S. (2015a). Glyphosate, pathways to modern diseases IV: cancer and related pathologies. J. Biol. Phys. Chem. 15, 121–159. doi: 10.4024/11SA15R.jbpc.15.03
Samsel, A., and Seneff, S. (2015b). Glyphosate, pathways to modern diseases III: manganese, neurological diseases, and associated pathologies. Surg. Neurol. Int. 6:45. doi: 10.4103/2152-7806.153876
Séralini, G. E., Clair, E., Mesnage, R., Gress, S., Defarge, N., Malatesta, M., et al. (2014). Republished study: long term toxicity of a Roundup herbicide and a Roundup-tolerant genetically modified maize. Environ. Sci. Eur. 26, 14. doi: 10.1186/s12302-014-0014-5
Smith, K., Evans, D. A., and El-Hiti, G. A. (2008). Role of modern chemistry in sustainable arable crop protection. Philos. Trans. R. Soc. B Biol. Sci. 363, 623–637. doi: 10.1098/rstb.2007.2174
Székács, A., and Darvas, B. (2012). “Forty years with glyphosate,” in Herbicides -Properties, Synthesis and Control of Weeds, ed M. N. Hasaneen (InTech) Availablie online at: http://www.intechopen.com (Accessed March 2016).
Then, C. (2009). Risk assessment of toxins derived from Bacillus thuringiensis-synergism, efficacy, and selectivity. Environ. Sci. Pollut. Res. 17, 791–797. doi: 10.1007/s11356-009-0208-3
Then, C. (2013). High Levels of Residues from Spraying with Glyphosate found in Soybeans in Argentina. TESTBIOTECH Background Report. Available onlie at: http://www.testbiotech.de/sites/default/files/TBT_Background_Glyphosate_Argentina_0.pdf (Accessed March, 2016).
US EPA (1983). Life Long Feeding Study in Sprague-Dawley rat. Caswell Case File #661/524-308. Results and Discussion of a Submitted Monsanto Study. Washington, DC: Internal memoranda and evaluations February – May 1983, extracted from the archives of The United States Food and Drug Administration, Office of Pesticides and Toxic Substances.
US EPA (1985). Memorandum: Use of Historical Data in Determining the Weight of Evidence from Kidney Tumor Incidence in the Glyphosate Two-Year Feeding Study; and Some Remarks on False Positives. Washington, DC: EPA registry #524-308 two-year feeding study in mice. Internal document dated 26. February 1985 extracted from the archives of The United States Food and Drug Administration, Office of Pesticides and Toxic Substances.
US EPA (1993). Reregistration Eligibility Decision (RED) Glyphosate. Washington, DC: United States Environmental Protection Agency, Office of Prevention, Pesticides and Toxic Substances.
USDA (2016). Foreign Agricultural Service, Table 11: Soybeans: World Supply and Distribution. United States Department of Agriculture. Available online at: www.fas.usda.gov/psdonline
van Haver, E., Alink, G., Barlow, S., Cockburn, A., Flachowsky, G., Knudsen, I., et al. (2008). Safety and nutritional assessment of GM plants and derived food and feed: the role of animal feeding trials. Food Chem. Toxicol. 46, S2–S70. doi: 10.1016/j.fct.2008.02.008
Vazquez, M. A., and Nota, C. (2011). Report from the 1st national meeting of physicians in the crop-sprayed towns. Faculty of Medical Sciences, National University of Cordoba August 2010. Cordoba.
Viljoen, C. (2013). Letter to the Editor. Food Chem. Toxicol. 59, 809–810. doi: 10.1016/j.fct.2013.07.003
Walsh, L. P., McCormick, C., Martin, C., and Stocco, D. M. (2000). Roundup inhibits steroidogenesis by disrupting steroidogenic acute regulatory (StAR) protein expression. Environ. Health Perspect. 108:769. doi: 10.1289/ehp.00108769
Williams, G. M., Kroes, R., and Munro, I. C. (2000). Safety evaluation and risk assessment of the herbicide roundup and its active ingredient, glyphosate, for Humans. Regul. Toxicol. Pharmacol. 31, 117–165. doi: 10.1006/rtph.1999.1371
Zobiole, L. H. S., Kremer, R. J., Oliveira, R. S. Jr., and Constantin, J. (2011). Glyphosate affects chlorophyll, nodulation and nutrient accumulation of “second generation” glyphosate-resistant soybean (Glycine max L.). Pestic. Biochem. Physiol. 99, 53–60. doi: 10.1016/j.pestbp.2010.10.005
CrossRef Full Text | Google Scholar
Keywords: glyphosate safety-assessment, history of glyphosate-herbicides, chemical diversity of glyphosates, glyphosate tolerant transgenic crops, Roundup
Citation: Cuhra M, Bøhn T and Cuhra P (2016) Glyphosate: Too Much of a Good Thing? Front. Environ. Sci. 4:28. doi: 10.3389/fenvs.2016.00028
Received: 31 January 2016; Accepted: 29 March 2016;
Published: 28 April 2016.
Edited by:
Robin Mesnage, King’s College London, UK
Reviewed by:
Yearul Kabir, University of Dhaka, Bangladesh
Abdel-Tawab H. Mossa, National Research Centre, Egypt
Copyright © 2016 Cuhra, Bøhn and Cuhra. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Marek Cuhra, marek.cuhra@gmail.com